Mesothelioma, a rare and aggressive cancer primarily associated with asbestos exposure, has long posed significant challenges for medical professionals due to its poor prognosis and limited treatment options. With a median survival rate of only 12-16 months and a five-year survival rate below 10%, the need for innovative therapies has been critical. In recent years, Tumor Treating Fields (TTFields) have emerged as a groundbreaking approach in mesothelioma treatment, offering new hope to patients battling this devastating disease.
Approved by the FDA in 2019 for treating malignant pleural mesothelioma, TTFields represent a significant advancement in cancer therapy. This non-invasive treatment modality utilizes low-intensity electric fields to disrupt cancer cell division, targeting inoperable pleural mesothelioma when used in conjunction with chemotherapy. The development of TTFields marks a pivotal moment in mesothelioma care, as it is the first FDA-approved treatment since the 2004 approval of the chemotherapy regimen combining Alimta (pemetrexed) and cisplatin.
Clinical trials have demonstrated promising results, with patients receiving TTFields in combination with chemotherapy showing an average overall survival of 18.2 months, compared to 12.1 months for those receiving chemotherapy alone. Notably, patients with epithelial mesothelioma exhibited even more favorable outcomes, with an average survival of 21.2 months. These statistics underscore the potential of TTFields to significantly improve patient outcomes and quality of life in the fight against mesothelioma.
This article explores the current state of TTFields in mesothelioma treatment, its impact on patient care, challenges faced in implementation, and future directions for research and clinical practice. By examining the mechanisms of action, clinical effectiveness, and potential for personalized treatment approaches, we aim to provide a comprehensive overview of this innovative therapy and its role in advancing mesothelioma treatment strategies.
Current State of Tumor Treating Fields in Mesothelioma
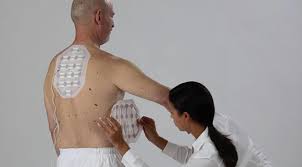
The implementation of Tumor Treating Fields (TTFields) in mesothelioma treatment has gained rapid acceptance in clinical practice. As of October 2019, multiple cancer centers across the United States have incorporated this therapy into their treatment protocols. The Optune Lua system, also known as NovoTTF-100L, delivers TTFields through four adhesive pads worn on the chest. Patients are instructed to use the device for 18 to 20 hours daily, allowing for a flexible lifestyle while receiving treatment.
Recent studies have shown that TTFields, when combined with chemotherapy, can extend survival rates for mesothelioma patients. A clinical trial reported a median survival of approximately 18.2 months for patients receiving this dual approach, compared to shorter survival times for patients treated with traditional chemotherapy alone. Notably, patients diagnosed with the epithelioid cell type achieved the longest survival durations, around 21.2 months.
Impact Analysis of TTFields on Mesothelioma Treatment
The introduction of TTFields has had a profound impact on mesothelioma treatment strategies and patient outcomes. Research indicates that TTFields not only enhance the effectiveness of chemotherapy but also lead to significant increases in DNA damage within cancer cells, promoting cell death. This dual-action approach has shown particular efficacy in epithelioid mesothelioma, the most common subtype.
TTFields therapy has a favorable side effect profile when compared to traditional chemotherapy. The most prevalent side effect reported is skin irritation at the site of electrode application, which is manageable with topical treatments. Only 4% of patients reported significant skin irritation, making TTFields an appealing option for patients seeking to maintain quality of life during treatment.
Challenges in TTFields Implementation for Mesothelioma
Despite its promising results, TTFields therapy faces several challenges. Access to treatment remains a concern, with only about 40 cancer treatment centers in the U.S. certified to prescribe this therapy as of 2019. The need for consistent daily use can be challenging for some patients, requiring a commitment to wear the device for 18-20 hours per day.
The effectiveness of TTFields appears to vary based on the histological subtype of mesothelioma, with sarcomatoid mesothelioma showing less responsiveness compared to epithelioid types. This variability highlights the need for personalized treatment approaches and further research into the mechanisms of action across different mesothelioma subtypes.
Future Directions in TTFields for Mesothelioma Treatment
Ongoing research is exploring combinations of TTFields with various chemotherapy regimens to optimize treatment efficacy. There is growing interest in understanding the molecular mechanisms behind TTFields’ effectiveness, particularly in different mesothelioma subtypes. Future studies aim to identify biomarkers that could predict patient response to TTFields, potentially leading to more personalized treatment approaches.
As of October 2019, TTFields were being implemented in multiple cancer centers across the United States, illustrating its rapid acceptance into clinical practice. Experts express optimism regarding TTFields as a viable option aimed at improving survival rates and offering an additional layer of treatment for patients grappling with mesothelioma. As accessibility improves and more oncologists become familiar with the therapy, TTFields are expected to play an increasingly significant role in comprehensive mesothelioma treatment plans.
Conclusion
Tumor Treating Fields represent a significant advancement in the treatment of mesothelioma, offering improved survival rates and quality of life for patients facing this challenging diagnosis. The therapy’s non-invasive nature, combined with its demonstrated efficacy, particularly in epithelioid mesothelioma, positions TTFields as a valuable addition to the limited arsenal against this aggressive cancer. With average overall survival extending to 18.2 months when combined with chemotherapy, TTFields have shown promise in extending life expectancy beyond traditional treatment methods.
As research continues and access expands, TTFields have the potential to transform the standard of care for mesothelioma patients. While challenges remain, including limited accessibility and varying effectiveness across mesothelioma subtypes, the integration of TTFields into treatment protocols marks a critical step forward. The ongoing exploration of TTFields in combination with other therapies and the pursuit of personalized treatment approaches offer hope for further improvements in patient outcomes. As we move forward, the role of TTFields in mesothelioma treatment is likely to evolve, potentially offering new avenues for combating this devastating disease and improving the lives of those affected.
References
- Ceresoli, G. L., et al. (2019). Lancet Oncology, 20(12), 1702-1709.
- Novocure. (2019). Optune Lua Patient Information and Operation Manual.
- Mun, E. J., et al. (2018). Cancer Management and Research, 10, 5641-5647.
- Stupp, R., et al. (2015). JAMA, 314(23), 2535-2543.
- FDA. (2019). FDA approves device for treatment of malignant pleural mesothelioma.
- American Cancer Society. (2022). Malignant Mesothelioma Survival Rates.
- Giladi, M., et al. (2015). Scientific Reports, 5, 18046.
- Weinberg, U., et al. (2021). Frontiers in Oncology, 11, 634752.
- Bianchi, C., & Bianchi, T. (2017). Indian Journal of Occupational and Environmental Medicine, 21(2), 82-85.