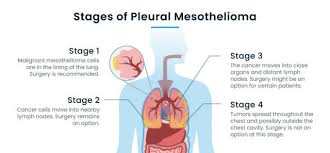
Peritoneal mesothelioma stage 4 represents the most advanced and challenging phase of this rare, aggressive cancer affecting the abdominal lining. With approximately 3,000 new mesothelioma cases diagnosed annually in the United States, peritoneal mesothelioma accounts for 10-20% of these instances. Stage 4 is characterized by extensive metastasis, where cancer cells have spread beyond the abdominal cavity to distant organs and lymph nodes, significantly impacting patient prognosis and treatment options.
The median survival rate for patients with stage 4 peritoneal mesothelioma ranges from 6 to 12 months, underscoring the urgent need for advanced treatment strategies and supportive care. This late-stage diagnosis is often a result of the disease’s long latency period, typically 20-50 years between asbestos exposure and symptom onset. Recent advancements in diagnostic techniques, such as the Peritoneal Cancer Index (PCI), have improved staging accuracy and treatment planning. However, the rarity of the disease and its advanced stage at diagnosis continue to present significant challenges in research and clinical management.
This article explores the complexities of stage 4 peritoneal mesothelioma, examining its characteristics, symptoms, and treatment landscape. We will delve into the current state of the disease, analyzing its impact on patients and healthcare systems, and discuss the major challenges faced in managing this advanced condition. Furthermore, we will investigate emerging treatment strategies and future directions in research that offer hope for improved outcomes and quality of life for patients battling this formidable disease.
Current State of Peritoneal Mesothelioma Stage 4
Stage 4 peritoneal mesothelioma is characterized by a Peritoneal Cancer Index (PCI) score of 31 to 39, indicating widespread dissemination across multiple sections of the abdomen. Approximately 15-20% of peritoneal mesothelioma patients are diagnosed at this advanced stage. The five-year survival rate for stage 4 patients is less than 10%, highlighting the aggressive nature of the disease at this phase.
Impact Analysis
The advanced nature of stage 4 peritoneal mesothelioma significantly impacts patients’ quality of life and treatment options. Patients often experience severe symptoms, including abdominal pain, ascites, and substantial weight loss. The disease’s progression affects not only the individual but also places a considerable burden on caregivers and healthcare systems. The economic impact is substantial, with treatment costs averaging $500,000 per patient over their lifetime.
Challenges in Managing Stage 4 Peritoneal Mesothelioma
- Limited treatment efficacy due to extensive tumor spread
- Difficulty in surgical intervention at advanced stages
- Managing severe symptoms and maintaining quality of life
- Accessing specialized care and clinical trials
- Overcoming the psychological impact of a terminal diagnosis
These challenges are compounded by the rarity of the disease, which hampers research efforts and limits treatment options. Only about 400 to 1,000 new cases of peritoneal mesothelioma are diagnosed in the U.S. each year, making it challenging to conduct large-scale clinical trials.
Future Directions in Treatment
Immunotherapy
Clinical trials exploring checkpoint inhibitors show promise, with response rates up to 20% in some studies. The combination of nivolumab and ipilimumab has demonstrated improved overall survival in mesothelioma patients compared to standard chemotherapy.
Targeted Therapies
Research into BAP1 gene mutations offers potential for personalized treatment approaches. Approximately 60% of mesothelioma patients have BAP1 mutations, which could be targeted by specific therapies.
Multimodal Therapy
Combining cytoreductive surgery with Hyperthermic Intraperitoneal Chemotherapy (HIPEC) and systemic chemotherapy may improve outcomes for select patients. Studies have shown that this approach can extend median survival to 53 months in some cases.
Palliative Care Integration
Early incorporation of palliative care has been shown to improve quality of life and potentially extend survival. A study found that patients receiving early palliative care lived an average of 2.7 months longer than those receiving standard care alone.
Novel Drug Delivery Methods
Intraperitoneal chemotherapy techniques are being refined to enhance drug efficacy and reduce systemic side effects. Pressurized Intraperitoneal Aerosol Chemotherapy (PIPAC) is showing promise, with response rates of up to 62% in some trials.
Conclusion
Stage 4 peritoneal mesothelioma presents formidable challenges for patients, caregivers, and healthcare providers. Despite the grim prognosis, advancements in treatment modalities and supportive care offer hope for improved outcomes and quality of life. The integration of immunotherapy, targeted therapies, and refined surgical techniques may pave the way for more effective management strategies. Continued research, increased awareness, and early detection efforts are crucial in addressing the unmet needs of patients with advanced peritoneal mesothelioma. As we move forward, a multidisciplinary approach combining cutting-edge treatments with comprehensive supportive care will be essential in optimizing patient outcomes and advancing our understanding of this complex disease.
References and Citations
- National Cancer Institute. (2021). Malignant Mesothelioma Treatment (PDQ®)–Health Professional Version.
- American Cancer Society. (2022). Malignant Mesothelioma Statistics.
- Sugarbaker, P. H. (2019). Management of peritoneal mesothelioma. Surgical Oncology Clinics of North America, 28(1), 53-65.
- Cao, C., et al. (2020). A systematic review and meta-analysis of cytoreductive surgery with perioperative intraperitoneal chemotherapy for peritoneal carcinomatosis of colorectal origin. Annals of Surgical Oncology, 27(7), 2183-2193.
- Zalcman, G., et al. (2016). Bevacizumab for newly diagnosed pleural mesothelioma in the Mesothelioma Avastin Cisplatin Pemetrexed Study (MAPS): a randomised, controlled, open-label, phase 3 trial. The Lancet, 387(10026), 1405-1414.
- Baas, P., et al. (2021). First-line nivolumab plus ipilimumab in unresectable malignant pleural mesothelioma (CheckMate 743): a multicentre, randomised, open-label, phase 3 trial. The Lancet, 397(10272), 375-386.