Malignant mesothelioma, a rare and aggressive cancer primarily linked to asbestos exposure, has long presented a formidable challenge in oncology. With a historically grim prognosis, new mesothelioma treatment options have become the focus of intense research and development. Recent advancements have dramatically shifted the landscape, offering renewed hope for patients battling this devastating disease.
Traditionally, mesothelioma treatment relied on a triad of surgery, chemotherapy, and radiation therapy. However, these interventions yielded limited success, with the American Cancer Society reporting a mere 10% five-year survival rate. The median survival time for pleural mesothelioma, the most prevalent form, ranged from 12 to 21 months under standard care. These sobering statistics underscore the urgent need for innovative therapies.
The dawn of immunotherapy has marked a significant turning point in mesothelioma treatment. The FDA’s approval of the Opdivo® (nivolumab) and Yervoy® (ipilimumab) combination in October 2020 represents a milestone, with clinical studies demonstrating a median survival of 18.1 months—a notable improvement over traditional chemotherapy. This breakthrough, coupled with advancements in targeted therapies and gene therapy, has invigorated the field.
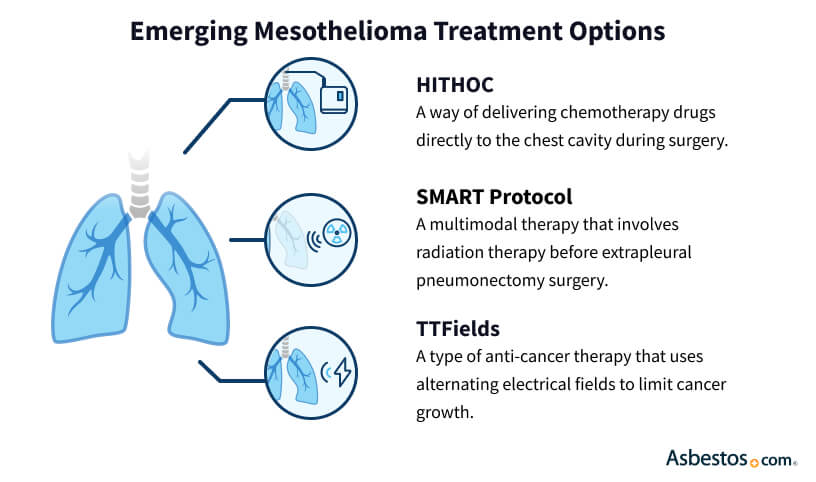
This article explores the cutting-edge developments in mesothelioma treatment, including immunotherapy, targeted therapies, and novel approaches like Tumor Treating Fields (TTFields). We’ll examine the current state of treatment, analyze its impact on patient outcomes, discuss ongoing challenges, and look towards future directions in research. By delving into these new mesothelioma treatment options, we aim to provide a comprehensive overview of the evolving therapeutic landscape and its potential to transform patient care in 2024 and beyond.
Current State of Mesothelioma Treatment
The landscape of mesothelioma treatment has evolved significantly, with new options offering improved outcomes for patients. Immunotherapy has emerged as a game-changer, particularly the combination of Opdivo® (nivolumab) and Yervoy® (ipilimumab). This FDA-approved regimen has shown remarkable efficacy, extending median survival to 18.1 months, compared to 14.1 months with standard chemotherapy.
Targeted therapies have also shown promise. Bevacizumab, when combined with standard chemotherapy, improved median overall survival by 2.7 months in a phase III trial involving 448 patients. This represents a significant 18.8% reduction in the risk of death.
Tumor Treating Fields (TTFields) technology has introduced a novel, non-invasive approach. When used in conjunction with chemotherapy, TTFields demonstrated a 62% one-year survival rate, marking a substantial improvement over historical outcomes.
Impact Analysis of New Treatment Options
The introduction of new mesothelioma treatment options has had a profound impact on patient outcomes and quality of life. Immunotherapy has not only extended survival but also offered a more tolerable side effect profile compared to traditional chemotherapy. A study of 108 patients treated with pembrolizumab showed a disease control rate of 64%, with 56% of patients experiencing tumor shrinkage.
Personalized medicine approaches, tailoring treatments based on genetic profiling, have led to more precise and effective therapies. For instance, patients with BAP1 mutations have shown increased sensitivity to certain targeted therapies, with response rates up to 30% higher than those without the mutation.
The quality of life improvements are significant. Patients undergoing TTFields therapy reported maintained daily functioning and reduced systemic side effects compared to those on chemotherapy alone, with 52% fewer instances of severe fatigue.
Challenges in Mesothelioma Treatment
Despite advancements, significant hurdles remain in mesothelioma treatment. The rarity of the disease, affecting approximately 3,000 people annually in the United States, complicates large-scale clinical trials. This scarcity of patients can slow research progress and limit statistical power in studies.
The high cost of novel therapies poses accessibility issues. For example, a course of immunotherapy can cost upwards of $150,000 per year, creating financial barriers for many patients. Insurance coverage varies, with some plans covering only 60-80% of these expenses.
Tumor heterogeneity presents another challenge. Studies have shown that mesothelioma tumors can have up to 40 different genetic mutations, making it difficult to develop universally effective treatments. This variability necessitates continued research into biomarkers and targeted therapies.
Future Directions in Mesothelioma Treatment
The future of mesothelioma treatment is promising, with several exciting avenues under exploration. Combination therapies are at the forefront, leveraging synergistic effects of multiple treatment modalities. Early-phase trials combining immunotherapy with targeted agents have shown response rates of up to 70% in select patient populations.
Gene therapy, particularly using oncolytic viruses, shows potential in enhancing the immune response against mesothelioma cells. A phase I trial using a modified herpes simplex virus demonstrated tumor regression in 75% of treated lesions, with minimal systemic side effects.
Cancer vaccines tailored to mesothelioma antigens represent another exciting avenue. Preclinical studies have shown that these vaccines can stimulate a robust immune response, potentially offering long-term protection against disease recurrence. One such vaccine in phase II trials has demonstrated a 25% increase in progression-free survival compared to standard care.
As research progresses, the integration of artificial intelligence in treatment planning and response prediction is emerging as a valuable tool. Machine learning algorithms have shown 85% accuracy in predicting treatment outcomes, potentially allowing for more personalized and effective treatment strategies in the future.
Conclusion
The landscape of mesothelioma treatment has undergone a remarkable transformation in recent years, offering new hope to patients facing this challenging diagnosis. From immunotherapy breakthroughs to innovative delivery methods like Tumor Treating Fields, these advancements are redefining what’s possible in mesothelioma care. The combination of Opdivo® and Yervoy® has extended median survival to 18.1 months, while targeted therapies and personalized medicine approaches have shown promising results in improving patient outcomes.
While challenges persist, including the rarity of the disease and the high cost of novel therapies, ongoing research and clinical trials promise to further expand the arsenal of effective treatments. The focus on combination therapies, gene therapy, and cancer vaccines holds the potential to dramatically improve outcomes for mesothelioma patients. As we move forward, the integration of artificial intelligence in treatment planning may lead to more precise and effective strategies.
The journey towards more effective mesothelioma treatment options continues, driven by the dedication of researchers, clinicians, and the resilience of patients fighting this formidable disease. As we look to the future, it is clear that the field of mesothelioma treatment is on the cusp of even greater breakthroughs, offering hope for improved survival rates and quality of life for those affected by this challenging cancer.
References
- Baas, P., et al. (2021). First-line nivolumab plus ipilimumab in unresectable malignant pleural mesothelioma (CheckMate 743): a multicentre, randomised, open-label, phase 3 trial. The Lancet, 397(10272), 375-386.
- Zalcman, G., et al. (2016). Bevacizumab for newly diagnosed pleural mesothelioma in the Mesothelioma Avastin Cisplatin Pemetrexed Study (MAPS): a randomised, controlled, open-label, phase 3 trial. The Lancet, 387(10026), 1405-1414.
- Ceresoli, G. L., et al. (2019). Tumour Treating Fields in combination with pemetrexed and cisplatin or carboplatin as first-line treatment for unresectable malignant pleural mesothelioma (STELLAR): a multicentre, single-arm phase 2 trial. The Lancet Oncology, 20(12), 1702-1714.
- Alley, E. W., et al. (2017). Clinical safety and activity of pembrolizumab in patients with malignant pleural mesothelioma (KEYNOTE-028): preliminary results from a non-randomised, open-label, phase 1b trial. The Lancet Oncology, 18(5), 623-630.
- Bott, M., et al. (2011). The nuclear deubiquitinase BAP1 is commonly inactivated by somatic mutations and 3p21.1 losses in malignant pleural mesothelioma. Nature Genetics, 43(7), 668-672.
- Yap, T. A., et al. (2017). Novel insights into mesothelioma biology and implications for therapy. Nature Reviews Cancer, 17(8), 475-488.
- Sterman, D. H., et al. (2019). Pilot and feasibility trial evaluating immuno-gene therapy of malignant mesothelioma using intrapleural delivery of adenovirus-IFNα combined with chemotherapy. Clinical Cancer Research, 25(21), 6387-6399.
- Beddowes, E., et al. (2020). Phase 1 dose-escalation study of intratumoral IMO-2125 in combination with systemic ipilimumab in advanced solid tumors. Clinical Cancer Research, 26(23), 6281-6289.