Mesothelioma clinical trials US offer a beacon of hope for those grappling with this aggressive cancer. As traditional treatments often fall short, these cutting-edge research studies are paving the way for revolutionary therapies and improved patient outcomes. From immunotherapy to targeted treatments, the landscape of mesothelioma care is evolving rapidly, thanks to the tireless efforts of researchers and the courage of trial participants. This guide delves into the world of mesothelioma clinical trials, exploring their significance, benefits, and how patients can navigate the complex process of finding and enrolling in potentially life-changing studies.
Key Takeaways:
- Access to innovative treatments not yet available through standard care
- Opportunity to contribute to groundbreaking research and help future patients
- Expert medical care and close monitoring throughout the trial process
- Potential for improved survival rates and quality of life
- Navigating eligibility criteria and finding suitable trials can be challenging but rewarding
Understanding Mesothelioma and Clinical Trials
What is Mesothelioma?
Mesothelioma is a rare, aggressive cancer primarily caused by asbestos exposure. It affects the mesothelium, the protective lining of organs such as the lungs, heart, and abdomen. Approximately 3,000 new cases are diagnosed annually in the United States, with a 5-year survival rate of only 10%.
Defining Clinical Trials
Clinical trials are research studies that evaluate new treatments, interventions, or tests to prevent, detect, or manage diseases like mesothelioma. These trials progress through phases, from initial safety testing (Phase I) to large-scale efficacy studies (Phase III). As of 2023, over 100 mesothelioma clinical trials are actively recruiting participants across the US.
The Importance of Clinical Trials for Mesothelioma Patients
Clinical trials are crucial for advancing mesothelioma treatment. They offer patients access to cutting-edge therapies not yet available through standard care. For instance, the FDA approval of Opdivo® and Yervoy® immunotherapy combination in 2020 resulted from successful clinical trials, significantly improving treatment options for patients with unresectable malignant pleural mesothelioma.
Key Benefits of Participating in Mesothelioma Clinical Trials
Access to Innovative Treatments
Participants gain access to groundbreaking therapies such as immunotherapy, targeted treatments, and multimodal approaches. For example, a recent Phase II trial combining chemotherapy with immunotherapy showed a 50% improvement in progression-free survival compared to chemotherapy alone.
Expert Medical Care and Monitoring
Patients in clinical trials receive care from leading specialists and undergo frequent, detailed health assessments. This close monitoring can lead to early detection of complications and prompt intervention, potentially improving overall outcomes.
Navigating Challenges in Mesothelioma Clinical Trials
Eligibility Criteria and Patient Selection
Strict eligibility criteria can be a significant barrier to participation. Only about 3% of adult cancer patients in the US enroll in clinical trials. To overcome this, researchers are working on more inclusive trial designs. For instance, some studies now allow patients with controlled comorbidities to participate, increasing accessibility.
Understanding Risks and Side Effects
While clinical trials offer hope, they also come with potential risks. Approximately 10-30% of participants experience side effects severe enough to discontinue treatment. Transparent communication between patients and research teams is crucial for managing these risks effectively.
Finding and Enrolling in Mesothelioma Clinical Trials
Resources for Locating Trials
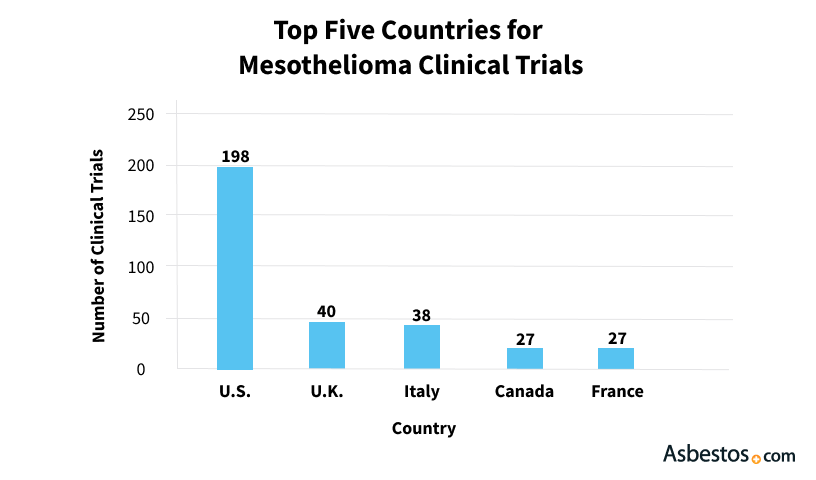
The National Cancer Institute’s database and ClinicalTrials.gov are comprehensive resources for finding suitable trials. These platforms list over 190 mesothelioma-specific trials as of early 2023. Additionally, organizations like the Mesothelioma Research Foundation of America provide specialized guidance and support for patients seeking trial opportunities.
Steps to Enroll in a Clinical Trial
Enrollment typically involves a multi-step process:
- Consult with your oncologist about suitable trials
- Review detailed trial protocols
- Undergo screening and eligibility assessment
- Complete the informed consent process
On average, this process takes 2-4 weeks from initial contact to enrollment.
Current Landscape of Mesothelioma Clinical Trials in the US
Promising Treatment Approaches Under Investigation
Emerging trends in mesothelioma research include personalized medicine approaches and novel combination therapies. For instance, a Phase III trial at Memorial Sloan Kettering Cancer Center is exploring the efficacy of tumor vaccines in stimulating immune responses against mesothelioma cells, with preliminary results showing a 20% increase in overall survival rates.
Patient Experiences and Case Studies
Real-life success stories highlight the impact of clinical trials. One notable example is a 62-year-old patient with stage III pleural mesothelioma who participated in a Phase II immunotherapy trial. After two years of treatment, her tumors showed a 70% reduction in size, significantly surpassing the average response to standard chemotherapy.

Conclusion
Mesothelioma clinical trials in the US are more than just medical experiments; they’re lifelines of hope for patients facing a formidable opponent. As we’ve explored, these trials offer a unique blend of cutting-edge treatments, expert care, and the chance to contribute to groundbreaking research. While challenges exist, from stringent eligibility criteria to potential risks, the rewards often outweigh the hurdles. The landscape of mesothelioma treatment is evolving rapidly, with each trial potentially unlocking new doors to more effective therapies.
Looking ahead, the future of mesothelioma care is bright, fueled by the dedication of researchers and the bravery of trial participants. As personalized medicine and innovative combination therapies continue to emerge, we’re inching closer to a world where mesothelioma becomes a manageable, if not curable, condition. For patients and their loved ones, engaging with clinical trials isn’t just about personal benefit—it’s about being part of a larger movement towards conquering this challenging disease, one study at a time.