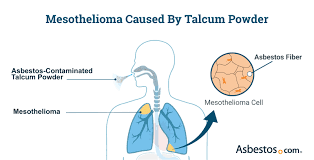
The connection between talcum powder and mesothelioma has emerged as a significant public health concern, challenging long-held assumptions about the safety of everyday cosmetic products. Recent studies have revealed a startling link between talcum powder use and the development of mesothelioma, a rare and aggressive cancer primarily associated with asbestos exposure. A comprehensive analysis of 166 mesothelioma cases found that 73.5% of patients reported talcum powder as their sole source of asbestos exposure, underscoring the potential dangers lurking in seemingly innocuous household items.
The global talc industry, valued at $1.9 billion in 2018, now faces unprecedented scrutiny as research uncovers the presence of asbestos in many talc-based products. This contamination stems from the geological proximity of talc and asbestos deposits, leading to potential cross-contamination during the mining process. Despite attempts by the U.S. Food and Drug Administration (FDA) to regulate talcum powder in the 1970s, industry pressure resulted in a retreat from stringent oversight, allowing manufacturers to self-test and report results.
The long-term health implications of talcum powder use are further complicated by the extended latency period between exposure and disease onset. With an average of 52.4 years from initial talc exposure to mesothelioma diagnosis, establishing direct causation becomes a formidable challenge. This article explores the scientific evidence linking talcum powder to mesothelioma, examines the legal ramifications faced by manufacturers, and discusses the broader public health implications of this emerging crisis.
Current State of Talcum Powder and Mesothelioma Research
Recent studies have intensified the debate surrounding the safety of talcum powder and its potential link to mesothelioma. In 2019, an FDA study found asbestos in 17.3% (9 out of 52) of cosmetic talc products tested, raising significant concerns about consumer safety. Johnson & Johnson, a major manufacturer of talc-based products, currently faces over 60,000 lawsuits related to its talcum powder products. In response to mounting pressure, J&J made the unprecedented decision in 2020 to discontinue talc-based baby powder sales in North America.
The World Health Organization has classified asbestos-contaminated talc as “definitely carcinogenic,” while the International Agency for Research on Cancer lists talc containing asbestos as a Group 1 carcinogen. These classifications underscore the severity of the health risks associated with contaminated talcum powder products.
Impact Analysis of Talcum Powder Use and Mesothelioma
The link between talcum powder and mesothelioma disproportionately affects women, who have historically been the primary users of these products for personal hygiene. A comprehensive study of 166 mesothelioma patients revealed that 65.7% had pleural mesothelioma, while 31.3% had peritoneal mesothelioma. This data challenges previous assessments that downplayed women’s susceptibility to asbestos-related diseases.
The economic impact of this health crisis is substantial. In June 2024, Johnson & Johnson proposed a $700 million settlement to resolve claims from multiple states regarding misleading marketing practices related to their talcum powder products. This significant financial liability highlights the far-reaching consequences of the talcum powder-mesothelioma connection.
Challenges in Addressing the Talcum Powder-Mesothelioma Link
One of the primary challenges in addressing the talcum powder-mesothelioma link is the long latency period of mesothelioma, which averages 52.4 years from initial talc exposure to diagnosis. This extended timeframe complicates efforts to establish direct causation and hinders timely regulatory responses.
Additionally, the lack of mandatory testing requirements for talc products allows potentially contaminated items to reach consumers. The absence of clear labeling requirements further exacerbates the issue, leaving consumers unaware of potential risks associated with talcum powder use.
Future Directions in Talcum Powder Safety and Mesothelioma Prevention
To address these challenges, stricter regulations and improved testing protocols are necessary. Advocates call for mandatory asbestos testing in all talc-based products and clear labeling of potential risks. Research efforts should focus on developing more sensitive detection methods for asbestos in talc, with a goal of identifying contamination at levels below the current detection limit of 0.1%.
Public health initiatives must educate consumers about the potential risks of talcum powder use and promote safer alternatives, such as cornstarch-based products. These efforts should target both consumers and healthcare providers to ensure widespread awareness of the potential link between talcum powder and mesothelioma.

Conclusion
The emerging link between talcum powder and mesothelioma represents a significant public health concern that demands immediate attention. As scientific evidence mounts and legal battles unfold, it is crucial for consumers, healthcare providers, and policymakers to remain informed about the potential risks associated with talcum powder use. The long latency period of mesothelioma, averaging 52.4 years, underscores the importance of proactive measures to prevent exposure. Moving forward, enhanced regulations, improved testing methods, and increased public awareness are essential to mitigate the health risks associated with talcum powder. By addressing these challenges, we can work towards ensuring consumer safety and preventing future cases of mesothelioma linked to talc exposure. The talcum powder industry must prioritize transparency and safety to regain public trust and prevent further health crises.
References and Citations
- International Agency for Research on Cancer. (2010). IARC Monographs on the Evaluation of Carcinogenic Risks to Humans, Volume 93: Carbon Black, Titanium Dioxide, and Talc.
- Moline, J., et al. (2020). Mesothelioma Associated with the Use of Cosmetic Talc. Journal of Occupational and Environmental Medicine, 62(1), 11-17.
- U.S. Food and Drug Administration. (2019). FDA Warns Consumers Not to Use Certain Cosmetic Products.
- Johnson & Johnson. (2020). Johnson & Johnson Consumer Health Announces Discontinuation of Talc-based Johnson’s Baby Powder in U.S. and Canada.
- World Health Organization. (2014). Chrysotile Asbestos.
- Rohl, A. N., & Langer, A. M. (1976). Identification and quantitation of asbestos in talc. Environmental Health Perspectives, 9, 95-109.
- Gordon, R. E., Fitzgerald, S., & Millette, J. (2014). Asbestos in commercial cosmetic talcum powder as a cause of mesothelioma in women. International Journal of Occupational and Environmental Health, 20(4), 318-332.