Immunotherapy drugs for mesothelioma are revolutionizing treatment options for this aggressive cancer. As medical science marches forward, two heavyweight contenders have emerged in the ring: Nivolumab (Opdivo) and Pembrolizumab (Keytruda). Both pack a punch against cancer cells, but which one takes the title belt? We’re diving into the nitty-gritty of these game-changing medications, comparing their strengths, potential drawbacks, and overall impact on patient care. Whether you’re navigating treatment options or simply curious about cutting-edge cancer therapies, buckle up for an enlightening journey through the world of mesothelioma immunotherapy. Get ready to explore how these drugs are reshaping the landscape of hope for patients worldwide.
Overview of Nivolumab (Opdivo)
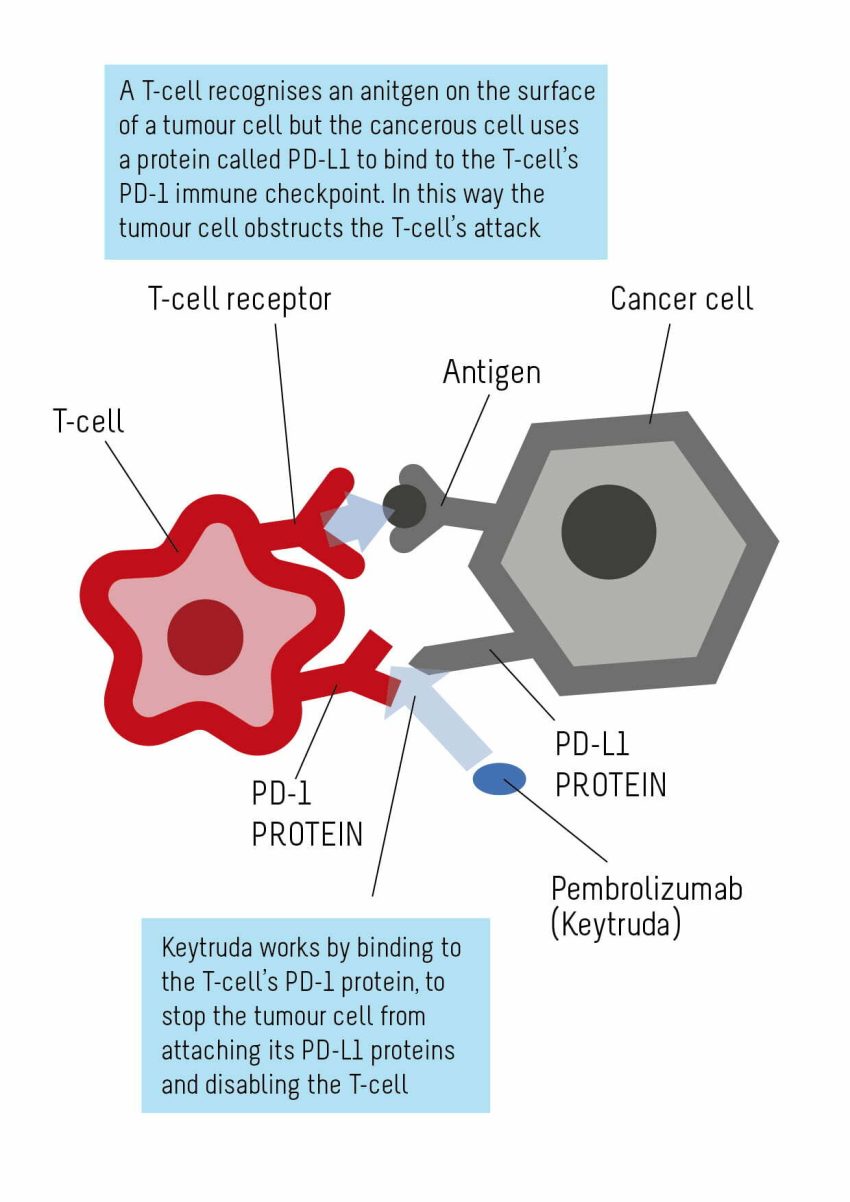
Nivolumab, marketed as Opdivo, is a groundbreaking immunotherapy drug that’s making waves in mesothelioma treatment. This PD-1 inhibitor works by blocking the PD-1 protein on T cells, effectively unleashing the immune system to attack cancer cells.
Key Features and Characteristics
- FDA-approved for first-line treatment of unresectable malignant pleural mesothelioma
- Administered intravenously every 2-4 weeks
- Often used in combination with Ipilimumab (Yervoy)
Advantages of Nivolumab
Clinical trials have shown impressive results for Nivolumab. The CheckMate 743 trial demonstrated a median overall survival of 18.1 months, compared to 14.1 months with chemotherapy alone. Moreover, the 2-year survival rate reached 41% for patients receiving Nivolumab plus Ipilimumab, versus 27% for chemotherapy.
Disadvantages of Nivolumab
While effective, Nivolumab isn’t without drawbacks. Common side effects include fatigue (reported in 37% of patients), rash (34%), and musculoskeletal pain (28%). The annual cost of treatment can exceed $150,000, posing potential accessibility issues for some patients.
Overview of Pembrolizumab (Keytruda)
Pembrolizumab, known by its brand name Keytruda, is another PD-1 inhibitor that’s shown promise in mesothelioma treatment. It works similarly to Nivolumab by enhancing the immune system’s ability to detect and destroy cancer cells.
Key Features and Characteristics
- FDA-approved for certain types of cancers with high tumor mutational burden
- Administered intravenously every 3-6 weeks
- Can be used as monotherapy or in combination with chemotherapy
Advantages of Pembrolizumab
Pembrolizumab has demonstrated efficacy in several studies. A phase 2 trial showed a disease control rate of 56% in previously treated mesothelioma patients. The median overall survival was 18 months, with some patients experiencing durable responses lasting over 3 years.
Disadvantages of Pembrolizumab
Side effects of Pembrolizumab can be significant, with 20-30% of patients experiencing severe adverse reactions. The most common side effects include fatigue (25%), nausea (20%), and decreased appetite (17%). Like Nivolumab, the cost of Pembrolizumab treatment is substantial, averaging around $150,000 per year.
Key Comparison Factors
Overall Survival Rates
Both drugs have shown improvements in overall survival compared to traditional chemotherapy. Nivolumab, when combined with Ipilimumab, demonstrated a median overall survival of 18.1 months, while Pembrolizumab monotherapy achieved a median of 18 months in previously treated patients.
Progression-Free Survival
The Nivolumab-Ipilimumab combination showed a median progression-free survival of 6.8 months, compared to 7.2 months for chemotherapy. Pembrolizumab demonstrated a median progression-free survival of 5.6 months in previously treated patients.
Quality of Life During Treatment
Both drugs generally offer better quality of life compared to chemotherapy. However, Nivolumab in combination therapy may lead to more immune-related adverse events (55%) compared to Pembrolizumab monotherapy (20-30%).
Side-by-Side Comparison
| Factor | Nivolumab (Opdivo) | Pembrolizumab (Keytruda) |
|---|---|---|
| Median Overall Survival | 18.1 months (with Ipilimumab) | 18 months (monotherapy) |
| FDA Approval Status | Approved for first-line treatment | Approved for certain cancer types |
| Common Side Effects | Fatigue (37%), Rash (34%) | Fatigue (25%), Nausea (20%) |
| Treatment Cost (Annual) | ~$150,000+ | ~$150,000 |
Both Nivolumab and Pembrolizumab represent significant advancements in immunotherapy drugs for mesothelioma. While they share similarities in their mechanism of action and overall efficacy, subtle differences in their approval status, side effect profiles, and combination therapy potential may influence treatment decisions. As research continues, these drugs are likely to play an increasingly important role in mesothelioma treatment strategies.
Conclusion
As we’ve seen, both Nivolumab and Pembrolizumab pack a punch in the fight against mesothelioma. They’re not one-size-fits-all solutions, though. Your best bet? Talk it out with your oncologist. They’ll weigh factors like your overall health, cancer stage, and personal preferences to tailor the right approach for you. Remember, these immunotherapy drugs are just part of the bigger picture. They might be combined with other treatments or used solo, depending on your unique situation. The world of mesothelioma treatment is evolving at breakneck speed, with new clinical trials and combination therapies popping up regularly. So, stay curious and keep the conversation going with your healthcare team. After all, the goal isn’t just to extend life, but to make those extra months or years count. Whether it’s Nivolumab, Pembrolizumab, or the next big breakthrough, the future of mesothelioma treatment is looking brighter than ever.