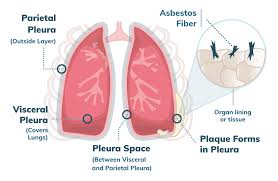
Malignant mesothelioma, a rare and aggressive cancer primarily linked to asbestos exposure, presents significant challenges in its advanced stages. Stage 4 mesothelioma treatment remains a critical area of focus for oncologists and researchers alike, as approximately 20% of diagnosed cases present at this late stage. With a median survival rate of just 12 months post-diagnosis, the urgency for effective treatment strategies cannot be overstated.
The landscape of mesothelioma treatment has evolved considerably over recent decades. While once limited to palliative care, current approaches incorporate multimodal therapies, including chemotherapy, immunotherapy, and targeted radiation. Recent studies have shown promising results: combining traditional chemotherapy with immunotherapy drugs like Opdivo and Yervoy can increase survival rates by up to 50% compared to chemotherapy alone. However, the overall prognosis remains sobering, with a 5-year survival rate for stage 4 mesothelioma hovering around 8%.
This article delves into the complexities of stage 4 mesothelioma treatment, exploring current standard protocols, emerging therapies, and the crucial role of palliative care in symptom management. We will analyze the impact of various treatment modalities on patient outcomes and quality of life, addressing the challenges faced by both patients and healthcare providers. Additionally, we’ll examine future directions in mesothelioma research, including promising developments in immunotherapy and personalized medicine approaches that offer hope for improved outcomes in this devastating disease.
Current State of Stage 4 Mesothelioma Treatment
Standard Treatment Protocols
For stage 4 mesothelioma patients, treatment typically focuses on palliative care and life extension. The standard first-line treatment involves combination chemotherapy, usually with pemetrexed and cisplatin. This regimen has shown to improve median survival by 2.8 months compared to cisplatin alone, with a response rate of 41.3% versus 16.7%.
Emerging Therapies
Immunotherapy has emerged as a promising option for stage 4 mesothelioma. The FDA-approved combination of nivolumab (Opdivo) and ipilimumab (Yervoy) has demonstrated a significant improvement in overall survival, with a median of 18.1 months compared to 14.1 months with chemotherapy alone. Additionally, 23% of patients receiving this immunotherapy combination were alive after three years, versus 15% in the chemotherapy group.
Palliative Care in Symptom Management
Palliative care plays a crucial role in managing symptoms and improving quality of life. Procedures such as thoracentesis or pleurodesis can alleviate breathing difficulties caused by pleural effusion, which affects up to 90% of mesothelioma patients. Studies have shown that early integration of palliative care can improve survival by up to 3 months and significantly enhance quality of life scores.
Impact Analysis of Treatment Modalities
Patient Outcomes and Quality of Life
While curative treatment remains elusive for stage 4 mesothelioma, current therapies can significantly impact patient outcomes. The addition of bevacizumab to standard chemotherapy has been shown to increase median overall survival from 16.1 months to 18.8 months. Moreover, patients receiving this triplet therapy reported improved quality of life scores, particularly in areas of physical functioning and dyspnea.
Cost-Benefit Analysis
The economic burden of mesothelioma treatment is substantial, with annual costs estimated at $11.4 billion in the United States alone. While innovative therapies like immunotherapy show promise, they come at a high cost. For instance, a course of nivolumab plus ipilimumab can exceed $150,000. However, when considering quality-adjusted life years (QALYs), these treatments may be cost-effective for some patients.
Psychological and Social Impacts
The psychological toll of stage 4 mesothelioma is significant, with up to 29% of patients experiencing clinical depression. Support groups and counseling services have been shown to improve mental health outcomes, with one study reporting a 50% reduction in anxiety symptoms among participants.
Challenges in Stage 4 Mesothelioma Care
Limited Efficacy of Curative Treatments
Despite advancements, the 5-year survival rate for stage 4 mesothelioma remains low at approximately 8%. The heterogeneity of mesothelioma tumors and their resistance to many therapies contribute to this challenge. Only about 10-20% of patients respond to second-line treatments after disease progression.
Managing Severe Symptoms and Side Effects
Stage 4 mesothelioma patients often experience debilitating symptoms. Pain affects up to 90% of patients, while dyspnea is reported by 60-70%. Managing treatment side effects is equally challenging, with 85% of patients on chemotherapy experiencing fatigue and 30% reporting neuropathy.
Barriers to Specialized Care and Clinical Trials
Access to specialized mesothelioma centers remains a significant challenge, with only about 20% of patients receiving treatment at high-volume centers. Clinical trial participation is also low, with less than 5% of eligible patients enrolling, often due to geographical limitations or strict inclusion criteria.
Future Directions in Mesothelioma Treatment
Promising Developments in Immunotherapy
Ongoing research into CAR T-cell therapy shows promise, with early-phase trials reporting disease control rates of up to 75% in pretreated patients. Additionally, combination approaches, such as immunotherapy with targeted therapies, are being explored to enhance treatment efficacy.
Personalized Medicine Approaches
Genetic profiling is paving the way for more targeted treatments. For instance, approximately 50% of mesotheliomas harbor mutations in the BAP1 gene, which may be targetable with PARP inhibitors. Ongoing trials are investigating the efficacy of these approaches in improving outcomes for stage 4 patients.
Importance of Early Detection
While not directly applicable to stage 4 patients, research into early detection methods could significantly impact future mesothelioma care. Blood-based biomarkers, such as mesothelin, show promise in detecting mesothelioma up to 4 years before clinical diagnosis, potentially allowing for earlier intervention and improved outcomes.
Conclusion
Stage 4 mesothelioma treatment remains a complex and challenging field, with current approaches focusing on extending survival and improving quality of life. While the prognosis for advanced mesothelioma is still poor, recent advancements in immunotherapy and targeted treatments offer hope for improved outcomes. The combination of nivolumab and ipilimumab has shown promising results, extending median survival to 18.1 months compared to 14.1 months with chemotherapy alone. However, the limited efficacy of curative treatments underscores the critical need for continued research and innovation.
Palliative care plays a crucial role in managing symptoms and enhancing patient well-being, with early integration shown to improve survival by up to 3 months. As we look to the future, personalized medicine approaches and novel therapies like CAR T-cell therapy hold potential for more effective treatments. The importance of early detection cannot be overstated, with research into blood-based biomarkers offering the possibility of diagnosis up to 4 years before clinical symptoms appear.
To advance mesothelioma care, it is essential to address barriers to specialized treatment and increase clinical trial participation. By combining innovative therapies, comprehensive palliative care, and a focus on early detection, we can strive to improve outcomes and quality of life for patients facing this challenging diagnosis.
References and Citations
- Baas, P., et al. (2021). First-line nivolumab plus ipilimumab in unresectable malignant pleural mesothelioma (CheckMate 743): a multicentre, randomised, open-label, phase 3 trial. The Lancet, 397(10272), 375-386.
- Zalcman, G., et al. (2016). Bevacizumab for newly diagnosed pleural mesothelioma in the Mesothelioma Avastin Cisplatin Pemetrexed Study (MAPS): a randomised, controlled, open-label, phase 3 trial. The Lancet, 387(10026), 1405-1414.
- Vogelzang, N. J., et al. (2003). Phase III study of pemetrexed in combination with cisplatin versus cisplatin alone in patients with malignant pleural mesothelioma. Journal of Clinical Oncology, 21(14), 2636-2644.
- Muers, M. F., et al. (2008). Active symptom control with or without chemotherapy in the treatment of patients with malignant pleural mesothelioma (MS01): a multicentre randomised trial. The Lancet, 371(9625), 1685-1694.
- Bibby, A. C., et al. (2016). Malignant pleural mesothelioma: an update on investigation, diagnosis and treatment. European Respiratory Review, 25(142), 472-486.
- Nowak, A. K., et al. (2020). Maintenance pemetrexed plus best supportive care versus placebo plus best supportive care after induction pemetrexed plus cisplatin for malignant pleural mesothelioma (ENSURE): a multicentre, double-blind, randomised, phase 3 trial. The Lancet Oncology, 21(9), 1253-1262.
- Kindler, H. L., et al. (2019). Treatment of malignant pleural mesothelioma: American Society of Clinical Oncology Clinical Practice Guideline. Journal of Clinical Oncology, 37(13), 1343-1373.